| CONTENT | REFERENCES | RESOURCES | SPONSORS | CONTRIBUTORS | HOW TO USE THIS CD |
| CLIMATE CHANGE IN CONTEXT | PREVIOUS | | | NEXT |
3.3.2.b Stratospheric Ozone Depletion
What is ozone? Where is ozone found? How is ozone both helpful and harmful? Why should we be concerned about a thinning ozone layer in our atmosphere? How does the thinning ozone layer change the amount of UltraViolet (UV) that reaches Earth?
Source: Solar UV in Our World (2002) ACER, Environment Canada, Health Canada www.acer-acre.org
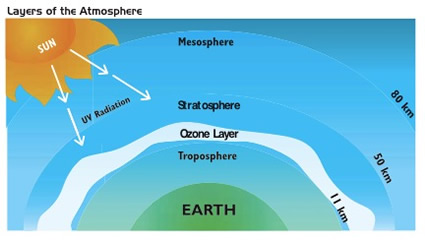
ACTIVITY 1
- Draw a vertical line to use as a bar graph with a scale to 100. Add the 3 layers of the atmosphere to scale. Calculate the ratio of each compared to the troposphere.
- Find a local map and overlay the line drawn in #1 to see the proportions as distances on the surface of the earth.
ACTIVITY 2 Research
1. How might STP be a factor in measuring on the earth’s surface. See DU below.
Solar UV in Our World (2002) ACER, Environment Canada, Health Canada www.acer-acre.org
ACTIVITY 3 Additional Activity
- Why are the molecules named in the last paragraph so dangerous in our atmosphere?
- Where are these catalytic molecules being produced?
- Research the production processes releasing N, Cl or Br to our atmosphere. Hint: Check out automobile emissions and include catalytic converters. See also Aerosols and Smog.
Source: Science and Impacts of Climate Change CD – Presentation Graphics (2002) MSC Environment Canada/ ESS Natural Resources Canada, December
ACTIVITY 4
- Where is the ozone layer in this photo taken from space?
- Measure the proportions of the 2 layers of the atmosphere seen here and compare the results to the work done on the first diagram.
The thin centre of the ozone layer in the stratosphere is often referred to as an ozone hole. The maps below show the relative thickness of the ozone layer over the poles.
Note the date (the time of year) on these maps.
What upsets the natural cycle of ozone in the stratosphere? The article explains how the pollutants from human activities break the ozone-oxygen cycle.
Chlorine is just one example of these catalytic molecules.
Source: Solar UV in Our World (2002) ACER, Environment Canada, Health Canada www.acer-acre.org
Source: Solar UV in Our World (2002) ACER, Environment Canada, Health Canada www.acer-acre.org
Correct the positioning of the arrows on the diagrams above using the color legend.
Source: Solar UV in Our World (2002) ACER, Environment Canada, Health Canada www.acer-acre.org
Source: Science and Impacts of Climate Change CD – Presentation Graphics (2002) MSC Environment Canada/ ESS Natural Resources Canada, December
ACTIVITY 5
- What year did the collection of satellite data begin?
- How does the ground-based data compare with the data colected from space?
- What is the general trend in concentration since 1965?
ACTIVITY 6 Research
Compare the Northern and Southern observations and explain the difference.
Hint: Check the information given above and in the graph below.
Source: Solar UV in Our World (2002) ACER, Environment Canada, Health Canada www.acer-acre.org
UV and ground-level ozone help make smog.
3.3.2d
Ground level pollution
Ozone and Smog
Ozone is toxic and active. It is a molecule made of 3 oxygen atoms held together with weak bonds that are ready to break. When ozone breaks apart it releases a charged atom of oxygen. The charged oxygen atom then immediately seeks to form a bond with other atoms or molecules. See the chemical reactions figure after the maps.
ACTIVITY 7
- What is the present ozone concentration across Ontario?
- Draw a map to the same scale as the one above.
- Plot only the changes since 1993. Use it as your base line for comparison.
Solar UV in Our World (2002) ACER, Environment Canada, Health Canada www.acer-acre.ca
Source: Solar UV in Our World (2002) ACER, Environment Canada, Health Canada www.acer-acre.org
ACTIVITY 8
- Check the raw materials side of the equation above and the major source of each.
- State the proper name and formula for each molecule in the entire equation.
- Why is there a time lag ?Answer the questions in the above exercise.
- State the time of day that pedestrians, cyclists, joggers, and dog walkers would be least exposed to smog. Most exposed?
sdsdsad